Testing, 1, 2, 3
Exploring the impact of cancer genomic profiling on insured mortality
August/September 2019We are in the midst of a genetic revolution. Advances in genomics have implications on pricing, underwriting, claims adjudication and product development. Offering comprehensive cancer genomic profiling to the insured post-policy issuance is a low-risk and affordable add-on feature that could benefit both the client and the insurance company’s profitability.
All insurers need a genomics strategy to guide them through these changing times. To date, most insurers have been more concerned about the potential negatives associated with genetic testing and adverse selection instead of looking at the positives that could be gained for both the public and for insurance companies. This article will discuss the benefits of analyzing the genetics of tumor cells, as this testing can help oncologists treat cancer patients using principles of precision oncology and can improve cancer outcomes. This should help to lower insured mortality and enhance client engagement.
Using the numbers discussed in this article, actuaries can calculate a possible return on investment (ROI) associated with insurance companies offering cancer genomic profiling to their insured clients.
What is Comprehensive Cancer Genomic Profiling?
Cancer is a genetic disease caused by mutations and other genomic alterations. Most mutations develop throughout life from exposures to carcinogens such as smoking, ultraviolet (sun) radiation, chemicals and viral infections. Inherited genetic mutations acquired at birth are responsible for 5 percent to 20 percent of all cancers.
Cancer genomic profiling analyzes a patient’s cancer, looking for various types of genetic alterations that are driving malignant transformation and tumor growth. Studies have shown that 37 percent to 49 percent of cancers have actionable genetic alterations where there already exists a targeted therapy to treat cancers that harbor these specific genetic mutations.1,2
Oncologists already use single-gene “companion diagnostic” genetic testing of cancer tissue, as these tests are linked to the use of U.S. Food & Drug Administration (FDA)-approved targeted therapies and immunotherapies used to treat specific types of cancer. Finding the same genetic mutation in a different type of cancer might allow the oncologist to get the patient into a clinical trial or use a targeted off-label medication to treat the patient.
Broader and more comprehensive cancer genomic profiling using next-generation sequencing interrogating 300+ cancer-related genes and gene byproducts is starting to be accepted in clinical practice. A recent study reported that 84 percent of cancer patients were found to have clinically relevant genomic alterations detected by comprehensive cancer genomic profiling that conventional testing did not identify.3
Currently, most commercial health insurers don’t cover comprehensive cancer genetic profiling, which can cost more than $5,800. One study of lung cancer patients showed 41 percent of patients did not follow the recommended clinical guidelines for genetic testing. The study mentions uncertainty regarding cost reimbursement as one of the barriers to ordering these tests.4 This is where genomic-based insurance products can play an important role at the time of cancer diagnosis to ensure access and coverage for these tests.
Advances in Genetics Are Lowering Cancer Mortality
Tremendous strides are being made in treating cancer. In the United States, cancer death rates have fallen 1.4 percent to 1.8 percent per year from 2006 to 2015.5 Cancer five-year survival also has improved. These favorable trends stem from improved cancer prevention and screening, as well as better treatments.
The number of targeted drugs and immunotherapies approved by the FDA for treating cancer is increasing, and each year a new cohort of clinical trials advancing these treatments is added to the queue.
Today, roughly 15 percent of patients with late-stage metastatic cancer undergo comprehensive cancer genomic profiling. Typically, this testing is done after current standard-of-care therapies have failed to stem the spread of cancer.
From the patient’s perspective, the prospect of a longer, healthier life is a clear positive. For the life insurer, this provides positive customer engagement and the chance to be viewed as part of a valuable solution for its customer. Life insurers also stand to benefit financially from increased use of this testing through the increased survival of the insureds affected.
Currently, there are no studies in the insured population to directly determine the mortality impact of offering cancer genomic profiling to a block of insured lives compared to the status quo. We have attempted to model these calculations using medical studies to derive key assumptions.
Three critical questions need to be answered in order to quantify the potential ROI for a life insurer that offers cancer genomic profiling to its clients:
- Which cancer patients could experience a survival benefit from getting comprehensive cancer genomic profiling?
- What percentage of advanced cancers receive genomically matched therapies?
- What is the survival benefit associated with the use of genomically matched cancer treatments?
It is important to recognize that, while we are attempting to answer these questions for the current point in time, it is likely that the numbers and percentages that answer these questions will increase over time. This will clearly support an appropriate “test and then expand” approach to the issue for life insurers/reinsurers.
Benefits of Comprehensive Cancer Genomic Profiling
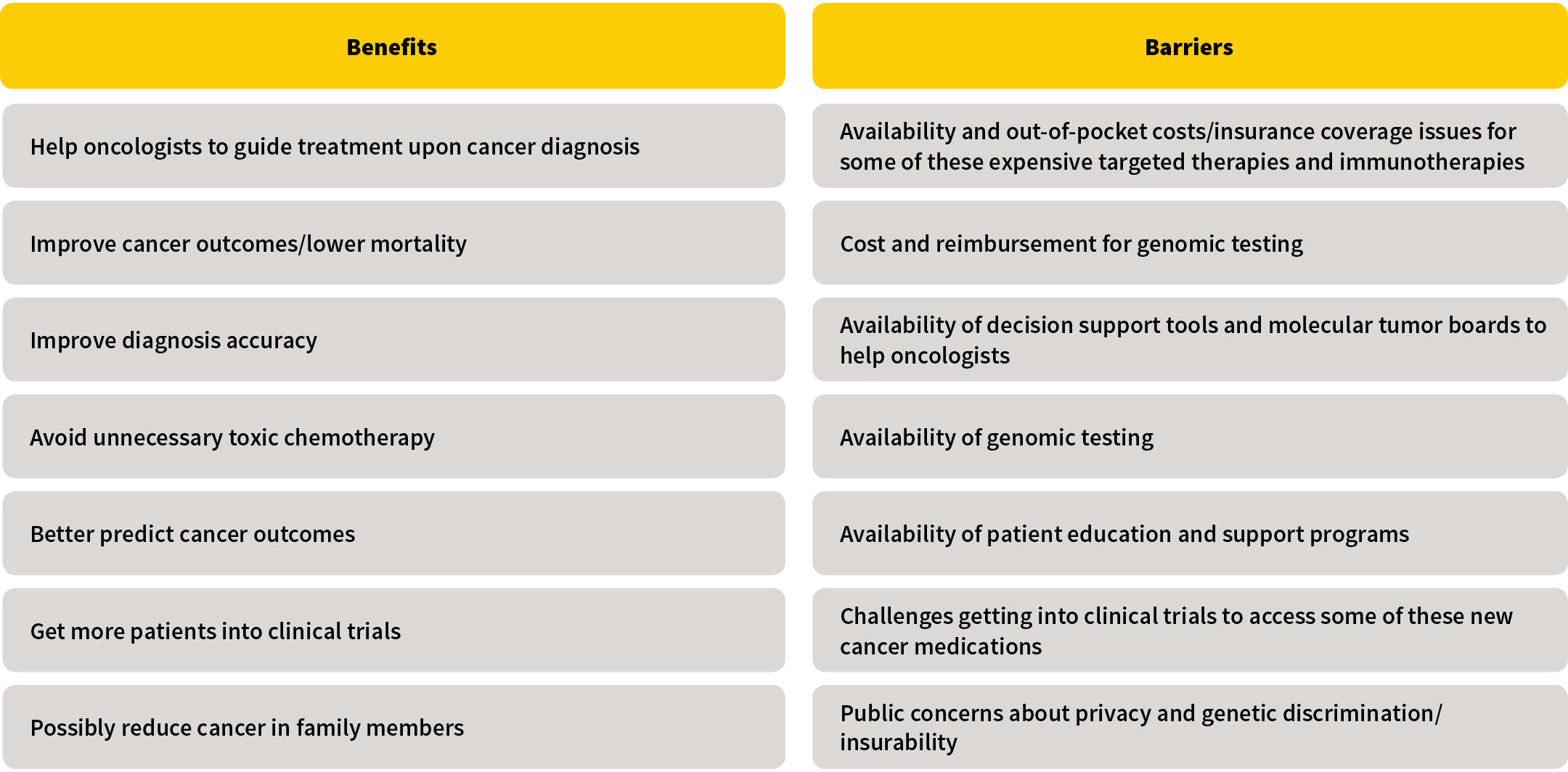
Most cancer patients can benefit in some way from getting their cancer genetically profiled as shown in Figure 1. Not only could profiling help guide treatment, but it also could lead to improved outcomes, benefitting both public health overall as well as the mortality in an insurance block of business.
Figure 1: Potential Benefits and Barriers to the Use of Comprehensive Cancer Genomic Profiling
Which Cancer Patients Get the Most Survival Benefit?
The cancer patients who would benefit most from comprehensive cancer genomic profiling are those who have either been found to have advanced stage cancer at the time of diagnosis or who have progressed from earlier stages of cancer to distant metastatic spread cancer.
We know that currently, the five-year relative survival rate for all cancers is 65 percent.6 We can assume almost all of the 35 percent of patients who die from their cancer will have distant spread metastatic disease.
Derived a different way, we know about 25 percent of all cancers are diagnosed when the cancer already has spread to distant sites.7 We then need to add for those patients who were unstaged who have distant spread cancer and also add for those patients who progress from earlier stages of cancer to metastatic disease.
We can surmise from these statistics that about 30 percent of cancers are advanced stage, and patients could possibly experience a survival benefit from getting comprehensive cancer genomic profiling performed.
What Percentage of Advanced Cancers Get Treated With Genomically Matched Therapies?
Not every patient with an actionable mutation will get on a targeted therapy because of various adverse factors such as becoming too sick to get any form of therapy.
Studies show 15 percent to 36 percent of advanced-stage cancer patients who get cancer genomic profiling receive targeted cancer therapies matched to the genetics of their cancer, and this percentage is increasing over time.8,9
We also need to take into account that comprehensive cancer genomic profiling can lead to treatment with immunotherapies, because one of the FDA indications to treat advanced solid tumors with immunotherapy relates to a particular genomic abnormality called mismatch repair deficiency, or tumors with high microsatellite instability.
In summary, it seems reasonable to assume that about 25 percent of advanced cancer patients who get genetically tested are expected to receive some form of matched cancer treatment as determined by their cancer’s genomic profile.
What is the Survival Benefit Associated With the Use of Genomically Matched Cancer Treatments?
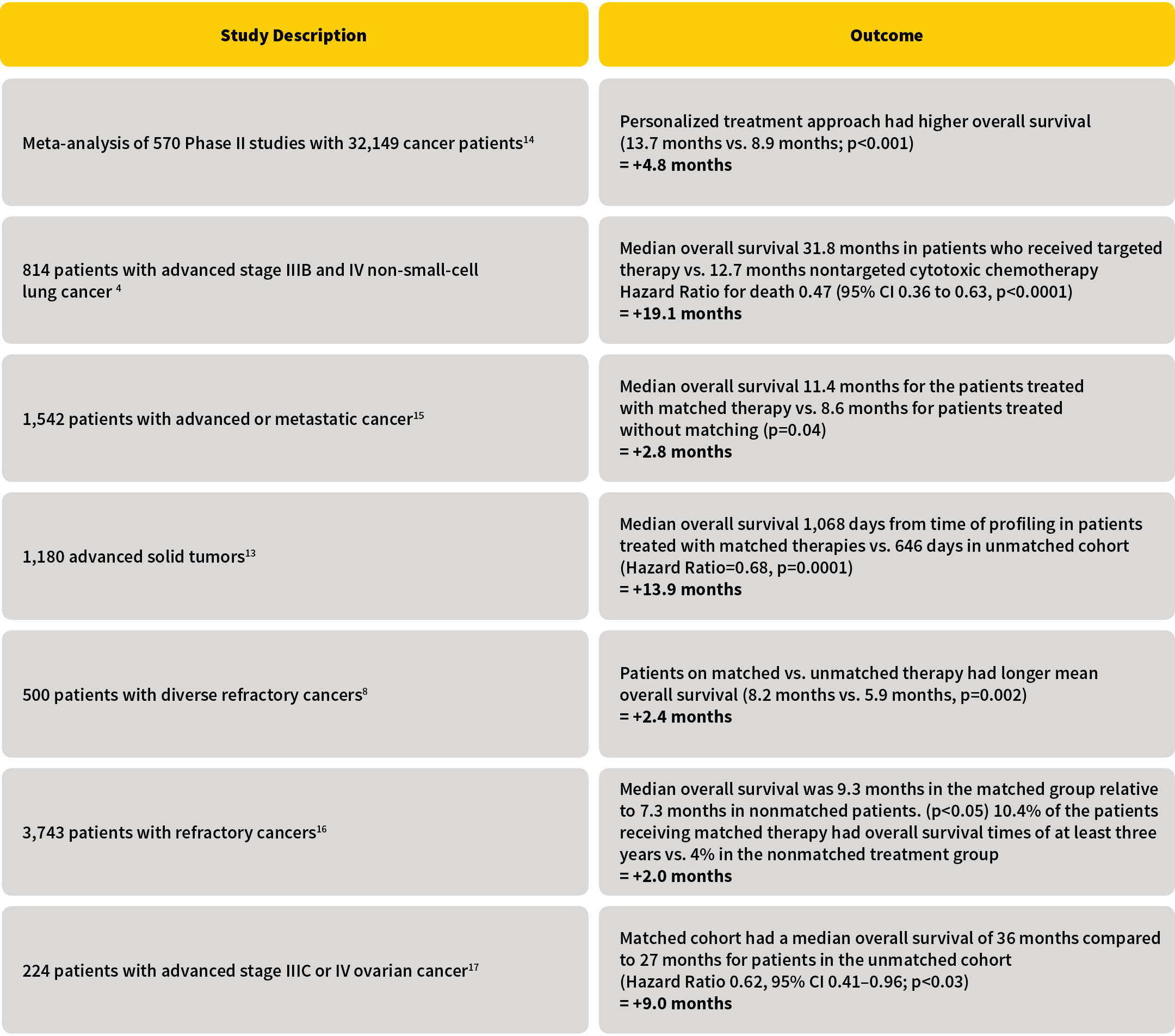
Many studies have shown that the use of genomically guided matched therapies improves patient outcomes.10,11,12,13,14,15,16,17,18,19 Using the medical literature to model an expected improvement in cancer mortality is challenging, because many of the medical studies have looked at different end points and use different types of cancer in their analysis. Figure 2 lists studies that report on median overall survival in advanced cancer patients who get treated with genomically matched therapies. These studies show that advanced cancer patients who receive matched therapies live on average two to 19 months longer compared with patients who are treated with unmatched therapies. The University of Texas MD Anderson Cancer Center’s Initiative for Molecular Profiling in Advanced Cancer Therapy (IMPACT) study analyzed 3,743 patients with refractory cancers and found that treatment with matched targeted therapy more than doubled the chance of living more than three years. (Results showed a 10.4 percent three-year survival rate in the matched therapy cohort vs. a 4 percent three-year survival rate in the nonmatched therapy cohort, p<0.0001).20
Figure 2: Survival Benefit Using Genetically Guided Therapies
As comprehensive cancer genomic profiling also leads to an increase in the use of immunotherapies, we need to add for the survival benefit of immunotherapy at least to some degree.
Combining mortality benefits associated with the use of all forms of genomically guided targeted therapies and immunotherapies, we can expect to see approximately a 10 month increase in median overall survival in advanced cancer patients who get treated with genomically matched therapies.
Potential Challenges to Consider With Cancer Genomic Profiling
Barriers to the adoption of cancer genetic testing in the community setting are listed in Figure 1.
Even though most physicians are very positive about the role cancer genomic profiling and precision medicine will play in oncology in the near future, some oncologists still debate the current clinical utility of comprehensive cancer genomic profiling, arguing that further research is needed and not all trials have shown statistically significant improved outcomes.21,22
Out-of-pocket cost and insurance coverage issues are also concerns. Comprehensive cancer genomic profiling leads to increased use of targeted therapies and immunotherapies that are expensive, costing upward of $12,500 per month. Some of these costs might not be covered by health insurers when these medications are used “off-label.” Fortunately, programs exist to help financially stressed patients who need these medications. In addition, new insurance product features such as accelerating part of the life sum assured could be designed to help pay for some of the treatment costs. Modeling research has shown this type of life insurance serious illness benefit rider might be cost effective to the insurer by capitalizing on the improved cancer survival.23
Potential technical challenges associated with the use of cancer genomic profiling also include:
- Inadequate cancer tissue sample for genetic analysis
- Ongoing genetic mutations in cancers
- Tumor heterogeneity within a cancer
Treating cancer patients earlier with genomically matched therapies and permitting repeat genetic testing remedies some of these challenges. In addition, the use of “liquid biopsies” is gaining significant research interest, as this type of blood testing might allow oncologists to follow a patient’s cancer genetics without the need for repeated tissue biopsies.
Public education is also important to avoid over-promoting the impact of cancer genomic profiling that could lead to overly optimistic public expectations in these advanced stage cancer patients.
Impact of Cancer Genomic Profiling on Health Care Costs
As comprehensive cancer genomic profiling is fairly new, the net impact on health care costs is unclear. This uncertainty is demonstrated in a 2018 Cardinal Health survey of 160 oncologists. Fifty-eight percent of those surveyed feel genomic tests will lead to health care cost savings in some cases. On a net basis, 69 percent of these oncologists believe genomic tests will increase oncology health care costs, but 31 percent feel genomic testing will either decrease or have no impact on health care costs.24
The cost of genetic testing leading to the use of expensive targeted therapies is mitigated at least in part by the cost savings attributed to being able to withhold conventional chemotherapy and possibly decrease hospitalization costs because some targeted therapies can be given orally at home. Some targeted therapies have fewer side effects than conventional chemotherapy, which could also lower health care spend. In addition, comprehensive cancer genomic profiling gets more patients into clinical trials. A recent study has shown that enrolling cancer patients into clinical trials could save the health insurer $25,000 per patient as drug costs are diverted to the study sponsor.25
Two small preliminary studies have shown that health care costs are about double in those patients who receive genetic testing guided targeted therapies, mostly due to longer treatment durations secondary to improved patient survival.26,27
Conclusion
By providing insured clients who develop cancer access to comprehensive cancer genomic profiling, the insurer stands to benefit directly from improved outcomes from a customer engagement, financial and public relations perspective. Those benefits are likely to increase as more genetically targeted treatments for cancer are developed and approved by the FDA.
Even applying a conservative set of assumptions for all of the topics discussed in this article, it appears that for policies and risks of a certain minimum size, a robust positive financial return can be achieved. A test case attempt focused on a block of policies of sufficient size can also serve to refine the assumptions and pinpoint how far down into an in-force block it makes sense to make these services available to insureds.
Dr. Smalley consults for Wamberg Genomic Advisors, which sells genomic products to employers and insurance companies.
References:
- 1. Zehir, Ahmet, Ryma Benayed, Ronak H. Shah, et al. 2017. Mutational Landscape of Metastatic Cancer Revealed From Prospective Clinical Sequencing of 10,000 Patients. Nature Medicine 23, no. 6:703–713. ↩
- 2. Massard, Christophe, Stefan Michiels, Charles Ferté, et al. 2017. High-throughput Genomics and Clinical Outcome in Hard-to-Treat Advanced Cancers: Results of the MOSCATO 01 Trial. Cancer Discovery 7, no. 6:586–595. ↩
- 3. Reitsma, Mitchell, John Fox, Pierre Vanden Borre, et al. 2019. Effect of a Collaboration Between a Health Plan, Oncology Practice, and Comprehensive Genomic Profiling Company From the Payer Perspective. Journal of Managed Care & Specialty Pharmacy 25, no. 5:601–611 ↩
- 4. Gutierrez, Martin E., Kelly Choi, Richard B. Lanman, et al. 2017. Genomic Profiling of Advanced Non-small Cell Lung Cancer in Community Settings: Gaps and Opportunities. Clinical Lung Cancer 18, no. 6:651–659. ↩
- 5. National Cancer Institute, Cancer Statistics. Cancer.gov, April 27, 2018 (accessed April 8, 2019). ↩
- 6. Siegel, Rebecca L., Kimberly D. Miller, and Ahmedin Jemal. 2019. Cancer Statistics, 2019. CA: A Cancer Journal for Clinicians 69:7–34. ↩
- 7. Ibid. ↩
- 8. Chawla, Anita, Filip Janku, Jennifer J. Wheler, et al. 2018. Estimated Cost of Anticancer Therapy Directed by Comprehensive Genomic Profiling in a Single-center Study. JCO Precision Oncology, 2:1–11. ↩
- 9. Albin, Nicolas, Anne McLeer, and Linda Sakhri. 2018. Precision Medicine: A Major Step Forward in Specific Situations, a Myth in Refractory Cancers? Bulletin du Cancer 105, no. 4:375–396. ↩
- 10. Supra note 2. ↩
- 11. Supra note 4. ↩
- 12. Supra note 8. ↩
- 13 Spetzler, David, Nick Xiao, Ken Burnett, et al. 2015. Multi-platform Molecular Profiling of 1,180 Patients Increases Median Overall Survival and Influences Treatment Decision in 53% of Cases. European Journal of Cancer 51, no. 3:S44. ↩
- 14. Schwaederle, Maria, Melissa Zhao, J. Jack Lee, et al. 2015. Impact of Precision Medicine in Diverse Cancers: A Meta-analysis of Phase II Clinical Trials. Journal of Clinical Oncology 33, no. 32:3817–38125. ↩
- 15. Tsimberidou, Apostolia-Maria, Sijin Wen, David S. Hong, et al. 2014. Personalized Medicine for Patients With Advanced Cancer in the Phase I Program at MD Anderson: Validation and Landmark Analyses. Clinical Cancer Research 20, no. 18:4827–4836. ↩
- 16. Tsimberidou, Apostolia-Maria, David S. Hong, Jennifer J. Wheler, et al. 2018. Precision Medicine: Clinical Outcomes Including Long-term Survival According to the Pathway Targeted and Treatment Period—The IMPACT Study. Journal of Clinical Oncology 36. ↩
- 17. Herzog, Thomas J., David Spetzler, Nick Xiao, et al. 2016. Impact of Molecular Profiling on Overall Survival of Patients With Advanced Ovarian Cancer. Oncotarget 7, no. 15:19840–19849. ↩
- 18. Schwaederle, Maria, Melissa Zhao, J. Jack Lee, et al. 2016. Association of Biomarker-based Treatment Strategies with Response Rates and Progression-free Survival in Refractory Malignant Neoplasms: A Meta-analysis. JAMA Oncology 2, no. 11:1452–1459. ↩
- 19. Kato, Shumei, Kellie Kurasaki, Sadakatsu Ikeda, and Razelle Kurzrock. 2018. Rare Tumor Clinic: The University of California San Diego Moores Cancer Center Experience With a Precision Therapy Approach. The Oncologist 23, no. 2:171–178. ↩
- 20. Supra note 16. ↩
- 21. West, Howard (Jack). 2016. No Solid Evidence, Only Hollow Argument for Universal Tumor Sequencing. JAMA Oncology 2, no. 6:717–718. ↩
- 22. Le Tourneau, Christophe, Jean-Pierre Delord, Anthony Gonçalves, et al. 2015. Molecularly Targeted Therapy Based on Tumour Molecular Profiling Versus Conventional Therapy for Advanced Cancer (SHIVA): A Multicentre, Open-label, Proof-of-Concept, Randomised, Controlled Phase 2 Trial. The Lancet Oncology 16, no. 13:1324–1334. ↩
- 23. Goldman, Dana P., and Darius Lakdawalla. Health Care’s Killer App: Life Insurance. The Wall Street Journal. March 20, 2019. ↩
- 24. Nabhan, Chadi. 2018. Oncology Insights: Views on Precision Medicine and Genomic Testing Practices From Specialty Physicians Nationwide. Cardinal Health Specialty Solutions. ↩
- 25. Supra note 3. ↩
- 26. Supra note 8. ↩
- 27. Haslem, Derrick S., S. Burke Van Norman, Gail Fulde, et al. 2017. A Retrospective Analysis of Precision Medicine Outcomes in Patients With Advanced Cancer Reveals Improved Progression-free Survival Without Increased Health Care Costs. Journal of Oncology Practice 13, no. 2:e108–e119. ↩
Copyright © 2019 by the Society of Actuaries, Chicago, Illinois.

